The origins of starch as an industrial material date back to Egyptian times when it was used in the manufacture of papyrus parchment and starch-based adhesive. Various other references exist to its use in Roman times as a whitener and stiffener for textiles. In Europe, the starch industry was mainly based on potatoes and evolved on the back of the textile industry where its use as a sizing agent was its main outlet. Post-war Europe realized that the seasonal nature of potato starch manufacture could be counterbalanced through the continuous milling of a maize crop. This in essence was the beginning of the diversification to the various starch sources which we encounter today.
Occurrence and appearance
As mentioned in The technology of starch production, starch is laid down in the tissues of many plants. The commercial realities of the starch recovery process however limit the industrial sources mainly to wheat, maize, potato and tapioca. The starch is found in granules whose shape and size are specific to the starch source. The starch source can be relatively easily identified from the extracted starch by viewing the granules under a medium-powered microscope. An iodine-based stain is commonly used for clarity and will also give further clues as to the amylose/amylopectin content of the granule. The granules vary from 2 to 150 μm in size, and are also characterized by specific shapes which range from round to polygonal and truncated. Figure 1.1 shows a range of the more common starches and Table 1.1 lists their particle size and structure type.
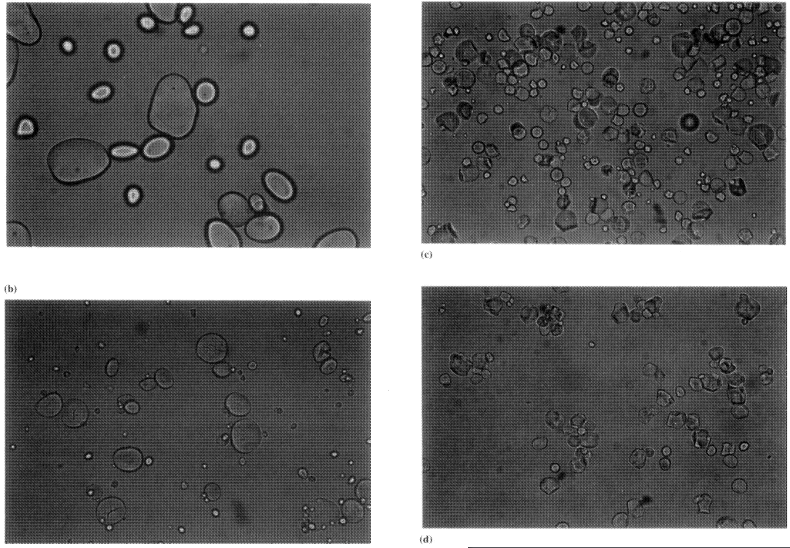
| Starch type | Diameter size range (μm) | Shape |
| Maize | 5-30 | Round, polyhedral |
| Wheat | 2-40 | Round |
| Potato | 50-150 | Ovoid |
| Tapioca | 4-30 | Kettle-shape |
Raw material composition
The composition of the raw material will be dependent on various factors, such as variety (of potato, maize etc.) and climatic conditions during growth and time of harvest. Storage conditions prior to processing may also influence minor component levels such as sugars. In the manufacture of starch for the production of glucose syrups, these will have little consequence except for perhaps pentosans and proteins which may lead to certain problems in processing and end use. Otherwise, adverse conditions of growth, harvest or storage of the raw material will only lead to reduced overall yields. Modern starch plants are designed to process a range of raw material qualities by making minor changes to the processing conditions. This is certainly not true for the manufacture of starch for its use per se or for the manufacture of products such as potato granules and flake, where the quality of the raw material and the fundamental composition of the starch may have pronounced effects on its efficacy in a given final manufacturing process, for example, in the manufacture of snack pellets. Table 1.2 lists the common starch sources and their typical composition.
| Source | Starch | Moisture | Protein | Fat | Fibre |
| Maize | 61 | 16 | 9 | 4 | 2 |
| Potato | 19 | 75 | 2 | 0.2 | 1.6 |
| Wheat | 65 | 14 | 13 | 2 | 3.5 |
| Tapioca | 30 | 63 | 3 | 0.3 | 5.5 |
Some interesting conclusions may be drawn from this basic table which gives some underlying criteria for raw material suitability for glucose production.
The starch content of the four raw materials on a dry weight basis is quite similar. However, when comparing the moisture contents of potato and tapioca starch (tuber starches) to those of wheat and maize starch (cereal starches) we can see a major difference. The tubers have considerably more moisture and the economics of production are therefore very different based on this fundamental difference. In Europe, for potatoes, a 30 mile radius is typically the limit of the harvest region owing to the unacceptable cost of transporting what is in effect 80% water. Potatoes are harvested on a campaign basis which usually lasts between August and January. The campaign may be extended providing no adverse weather conditions occur or if there is the possibility of adequate storage, usually underground. If the potato crop is subject to frost damage and subsequent spoilage it should be processed as soon as possible, being transformed into stable starch. This may be further processed into glucose syrup or stored as dried starch until required. The economics of storing the starch and its subsequent processing to glucose syrup are however not favorable because of the increased costs of producing dry starch.
Tapioca also has a high moisture content and is subject to even more regional growing restrictions around the starch plant than the potato since in the areas where it is grown there is usually a poor infrastructure which is often coupled to inadequate storage facilities. Furthermore, tapioca in the sub-tropical climate where it is grown is very susceptible to spoilage and must be processed almost immediately on arrival at the plant. If the processing does not commence within the first 24 h then it is necessary to dry the tuber to prevent spoilage.
The production of cereal starch on the other hand is well adapted to a continuous yearly production process. Both wheat and maize can be grown away from the immediate vicinity of the plant where they can be dried and stored quite favorably until required for processing. This gives the process a great deal of flexibility and further allows for material to be bought on the world spot markets to supplement production needs as required.
The protein content variation between tubers and cereals is quite significant. This protein content is usually expressed as Kjeldahl nitrogen X 6.25 and will include all nitrogen-containing compounds, such as any peptides, amides and amino acids that may be present. The starch recovery processes which will be discussed later are adapted to these botanical variations and maximize the yield of starch in its purest form.
The lipid fraction consists of triglyceride esters and free fatty acids (mainly palmitic, oleic and linoleic acids) together with some complex lipids (mainly phospholipids). The lipid content is substantially higher in cereals compared with tubers. The oil fraction occurs mainly in the germ of the cereal and is removed during the early stages of the starch manufacturing process.
The fiber content, as would be expected, is also higher in wheat and maize and primarily comes from the hull component. As with the germ it is mainly removed during the first stages of starch manufacture.
Starch composition
As previously discussed, starch consists of granules that are diverse in shape and size and are specific to their botanical source. The starch granule consists of two glucose polymer structures, namely amylose and amylopectin, and also contains moisture, lipids, proteins and mineral ions either present discretely in the matrix of the starch granule or as a specific complex with the starch polymer. Table 1.3 gives typical analyses for the common starches.
| Starch | Moisture | Lipid | Protein | Ash |
| Maize | 13 | 0.7 | 0.15 | 0.12 |
| Potato | 19 | 0.08 | 0.06 | 0.1 |
| Wheat | 14 | 0.8 | 0.3 | 0.15 |
| Tapioca | 13 | 0.2 | 0.2 | 0.2 |
Moisture
The moisture content of starch will vary depending on the surrounding conditions in which it is placed. Moisture contents are usually quoted at 65% relative humidity and 20oe, typical ambient conditions. Under these conditions, the moisture content of maize, wheat and tapioca is usually quoted at between 13-14%, and that of potato between 18-20%. Starches are usually supplied commercially at these equilibrium moisture contents to limit the risk of any dust explosions. The ability of starch to gain and lose large quantities of moisture is utilized in confectionery in starch-depositing processes.
Lipids
As can be seen in Table 1.3, higher lipid levels are found in cereal than in tuber/root starches. Lipids are of importance in the starch per se since they can form complexes with the starch polymers and make a significant contribution to the behavioral characteristics of the starch. Lipids can also impart off-flavors to a native or modified starch. These off-flavors can be significantly reduced by extensive washing of the starch in either native or modified form. Lipids have little significance as far as glucose syrup production is concerned.
Proteins
Both maize and wheat starch show a significantly higher residual protein level when compared to tapioca and potato starch. The proteinaceous matter is more important in a food starch application than in glucose syrup manufacture due to off-taints that may develop. These are referred to as specific ‘cereal flavor notes’ and are important in delicate-tasting foods, for example, instant desserts where a bland-tasting starch is a priority. Since the proteins and peptides have surface activity, of far more significance is the problem of foaming caused by these residues. If not removed they can contribute to unacceptable levels of browning and foaming during concentration or during subsequent use. These residual proteins can be reduced to acceptable levels through the correct use of enzymes and refining techniques. Furthermore, wheat starch consists of two distinct granular fractions, a small spherical fraction, 2-10 /-lm in diameter, and a second fraction consisting of large lenticular granules, 20-40 /-lm in diameter. Although the large granules only account for about 10% by number, they contribute about 90% of the weight of the starch. They also contain a relatively low level of protein compared with the small granules; 0.1 % compared with 1.5%. Several manufacturers now divide the starch stream into two granular fractions to take account of this.
Ash
All starches contain mineral ions. The cations are mainly sodium, potassium and magnesium but calcium, aluminium and iron are also found. Potato starch shows a significantly higher ash content than the other three major products. This higher level is a direct result of the high phosphate levels found in potatoes.
Amylose and amylopectin
Starch is not a homogeneous substance but is made up of two related but structurally different polymers, amylose and amylopectin. The starch granule has a biological nucleus around which the granule is built up. This is known as the hilum and may be concentric or eccentric depending on the botanical source. Three enzymes are thought to be involved in the process of granule development, phosphorylase, starch synthetase and branching enzyme. Table 1.4 gives the amylose and amylopectin content of different starches.
| Starch | Amylose (%) | Amylopectin (%) |
| Maize | 27 | 73 |
| Potato | 21 | 79 |
| Wheat | 27 | 73 |
| Tapioca | 14 | 86 |
The amylose content varies between 14% and 27% depending on the starch source although different varieties of the same source, e.g. maize, will have different amylose: amylopectin ratios. Specific starch varieties are now commercially available containing up to 99% amylopectin (the waxy starches) or up to 70% amylose (the high amylose starches). These starches have specific properties, are subject to growing restrictions and command a premium price. They would not normally be used in the manufacture of glucose syrups except perhaps as part of a reprocessing operation.
The basic building block of both amylose and amylopectin is the glucose monomer linked through the α-l,4 glycosidic bond (Figure 1.2). Another common way of describing the structures is that of polymeric condensation products of anhydroglucose units. During the development of the starch granule, there is a slight increase in the amylose-to-amylopectin ratio, with a commensurate increase in the molecular weights of both polymers. The cereal starches have a higher amylose content than the tuber starches.
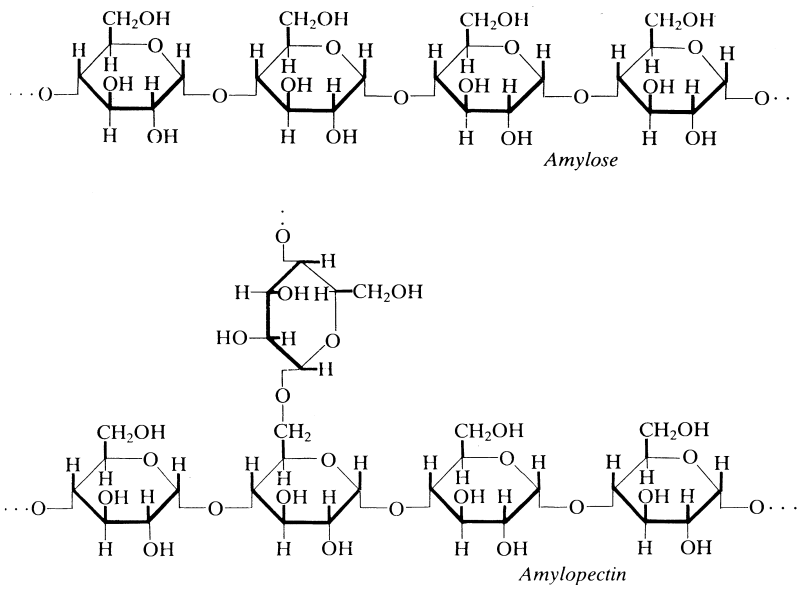
An indicative degree of polymerization (DP) of amylose is 102-103, with tuber starches showing in general a higher DP than the cereal starches. Amylopectin is usually considered as having a DP of an order of magnitude higher than amylose.
Various studies have shown that amylose is a straight-chain glucose polymer with a conformational helical structure, although there is evidence to suggest that a very small amount of branching may be present. Amylose has the ability to form inclusion complexes with iodine. The iodine molecule is held within a clathrate structure, with one iodine molecule per turn of the helix (six glucose units). The amylose-iodine complex has a characteristic blue colour. The colour and intensity observed are related to chain length (Table 1.5).
| Degree of polymerization | Color of starch/iodine complex |
| <12 | Yellow |
| <20 | Red |
| <30 | Purple |
| 30-40 DP | Mauve/blue |
| >40 DP | Blue |
Amylopectin is a highly complex branched molecule. The backbone of the molecule is still the (1-4) linked anhydroglucose unit but with branching points occurring at the carbon-6 primary alcohol group (Figure 1.2) every 10-12 glucose units. There is also occasional branching at carbon-3. The side chains off the main backbone are 20-30 glucose units in length. The molecular structure is not uniform and has areas of high branching and also of relative regularity. Starch granules consist of both crystalline and amorphous regions. Amylopectin occurs in both the amorphous and crystalline areas of the granule but is considered to be solely responsible for the crystallinity of starch. Amylose is associated with the amorphous regions of the granule.
When viewed under polarised light, starch granules show a birefringence pattern often referred to as a maltese cross as shown in Figure 1.3 for various starches. The center of the cross for a specific starch granule will be concentric or eccentric and will correspond to the hilum of the starch granule. These light diffraction patterns infer an eccentric or concentric radial organization to starch, originating at the hilum.
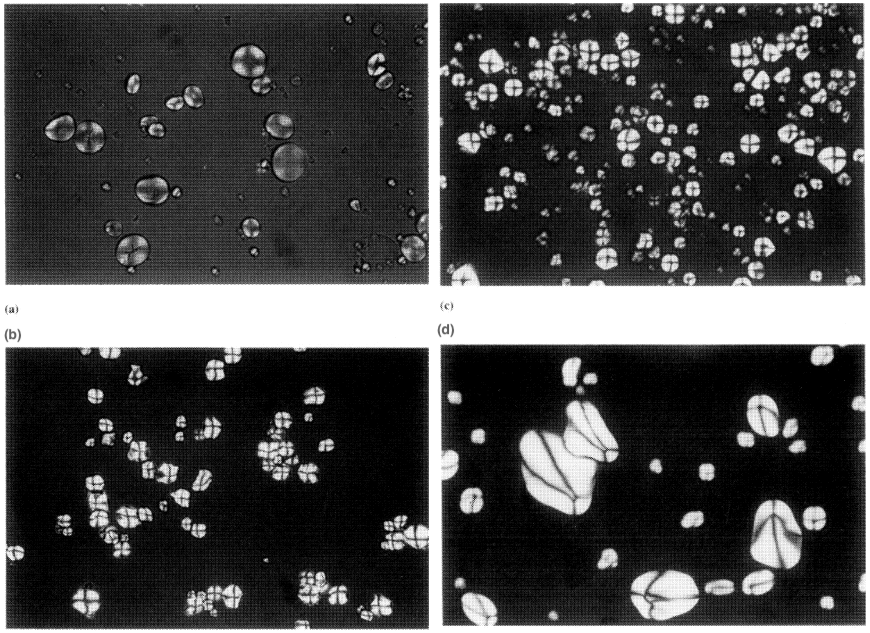
Different starches show birefringence patterns of varying strength indicating differing degrees of crystallinity in individual starches. This is shown in Figure 1.3.
Both intra- and inter-molecular hydrogen bonding are responsible for the structural integrity of the starch granule and also account for its insolubility in water. When a starch suspension is heated in water, heat overcomes the hydrogen bonding forces which hold the granule together allowing swelling or gelatinization of the starch granule to occur. This energy input is the first stage of both the acid and enzyme-catalyzed hydrolysis reactions since acid and enzymes have a little hydrolyzing effect on the ungelatinized starch.
